Introduction
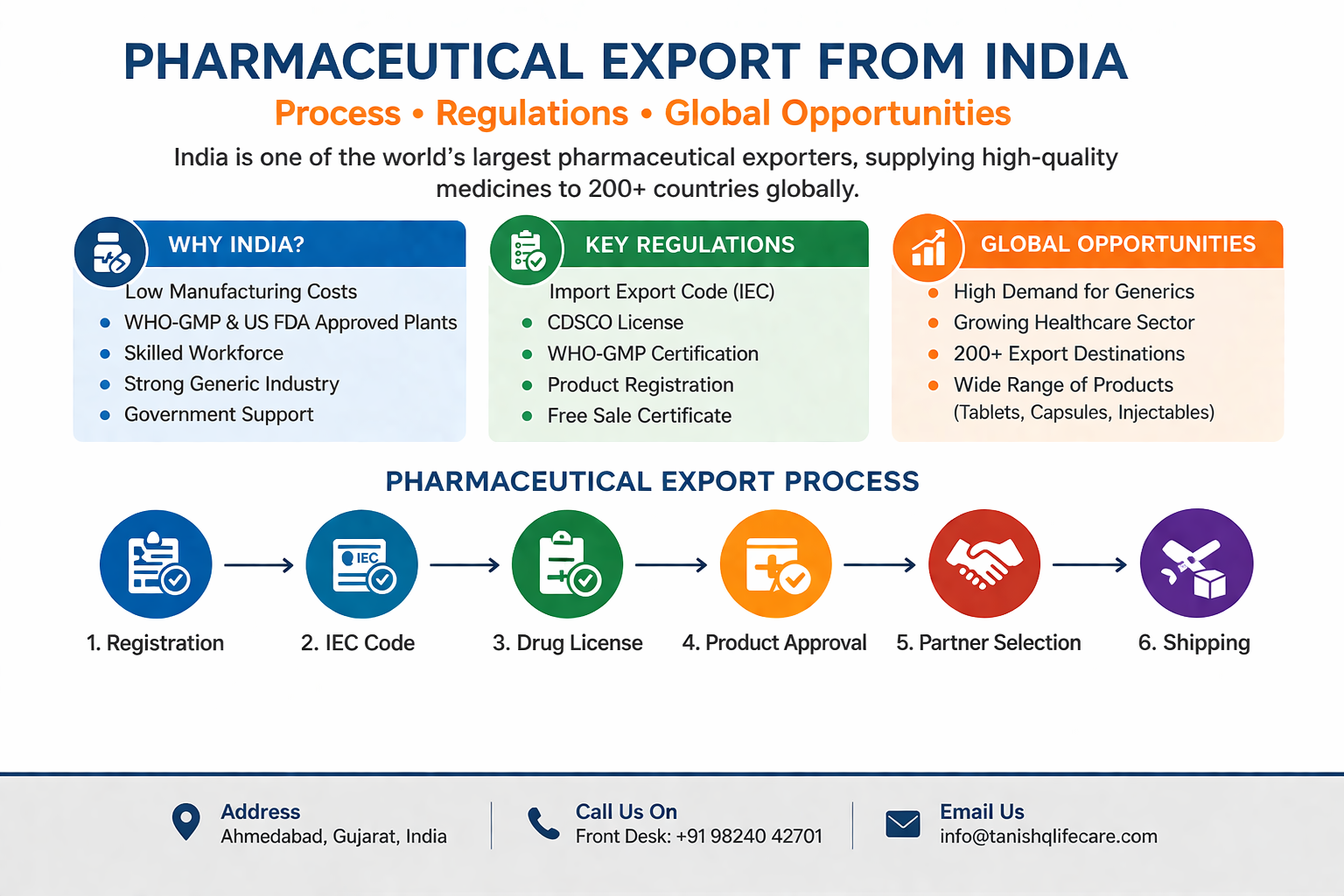
India has earned its place as the “pharmacy of the world,” supplying affordable and high-quality medicines across the globe. Over the past decade, pharmaceutical exports from India have witnessed exponential growth, driven by strong manufacturing capabilities, regulatory compliance, and increasing global demand for generic medicines.
Today, Indian pharmaceutical companies export to more than 200 countries, including highly regulated markets such as the United States, Europe, and emerging regions like Africa and Southeast Asia. This makes pharmaceutical export from India a highly lucrative opportunity for businesses looking to expand globally.
If you are planning to enter this dynamic sector, understanding the export process, regulatory requirements, and market opportunities is essential. In this comprehensive blog, we will walk you through everything you need to know about pharmaceutical export from India and how companies like Tanishq Life Care can help you succeed in this competitive industry.
What is Pharmaceutical Export?
Pharmaceutical export refers to the process of manufacturing medicines in one country and supplying them to international markets. These products include:
- Generic medicines
- Branded drugs
- Vaccines
- Nutraceuticals
- Active pharmaceutical ingredients (APIs)
Pharmaceutical export from India plays a crucial role in global healthcare by making affordable medicines accessible worldwide.
Why India is a Global Leader in Pharmaceutical Export
India’s dominance in the pharmaceutical export market is not accidental. It is the result of several strong advantages.
1. Cost-Effective Manufacturing
India offers significantly lower production costs compared to Western countries, allowing exporters to remain competitive in global markets.
2. High-Quality Standards
Indian pharmaceutical companies follow strict quality guidelines such as:
- WHO-GMP
- US FDA
- EU GMP
- ISO certifications
3. Strong Generic Medicine Industry
India is one of the largest producers of generic medicines, which are widely accepted across the globe.
4. Skilled Workforce
The country has a large pool of scientists, pharmacists, and healthcare professionals.
5. Government Support
The Indian government actively promotes pharmaceutical export through various schemes and policies.
Step-by-Step Process of Pharmaceutical Export from India
Understanding the export process is crucial for entering the global pharmaceutical market.
Step 1: Company Registration
Register your business as a legal entity (Private Limited, LLP, etc.).
Step 2: Obtain Import Export Code (IEC)
Issued by the Directorate General of Foreign Trade (DGFT), IEC is mandatory for export.
Step 3: Drug License
You must obtain a valid drug manufacturing or wholesale license from the appropriate authority.
Step 4: Product Registration
Register your pharmaceutical products in the target country as per their regulatory requirements.
Step 5: Appoint Export Agents or Distributors
Work with local partners in foreign markets to distribute your products.
Step 6: Documentation & Compliance
Prepare all required export documents (explained below).
Step 7: Logistics & Shipping
Arrange transportation through reliable logistics providers.
Key Regulations for Pharmaceutical Export from India
Pharmaceutical exports are highly regulated to ensure safety and quality.
1. CDSCO Approval
The Central Drugs Standard Control Organization regulates pharmaceutical products in India.
2. WHO-GMP Certification
Ensures compliance with international manufacturing standards.
3. Country-Specific Regulations
Each country has its own regulatory authority, such as:
- US FDA (USA)
- EMA (Europe)
- MHRA (UK)
4. Export NOC
In some cases, a No Objection Certificate (NOC) is required for export.
Documents Required for Pharmaceutical Export
Proper documentation is essential for smooth export operations.
Essential Documents:
- Import Export Code (IEC)
- Commercial Invoice
- Packing List
- Bill of Lading / Airway Bill
- Certificate of Origin
- Drug License
- Product Registration Certificate
- Certificate of Analysis (COA)
- Free Sale Certificate
Global Opportunities in Pharmaceutical Export
The global pharmaceutical market is expanding rapidly, creating massive opportunities for Indian exporters.
Key Growth Drivers:
- Increasing healthcare demand
- Aging population
- Rise in chronic diseases
- Demand for affordable medicines
India’s strength in generics and cost-effective production makes it a preferred partner worldwide.
Challenges in Pharmaceutical Export from India
While the opportunities are vast, exporters must also navigate challenges:
- Strict regulatory compliance
- Complex product registration processes
- Competition from global players
- Supply chain management
Working with experienced partners can help overcome these challenges efficiently.
How to Choose the Right Pharmaceutical Export Partner
Choosing the right partner can significantly impact your success.
Key Factors:
- Experience in global markets
- Regulatory expertise
- Product quality standards
- Certifications and approvals
- Reliable supply chain
- Transparent communication
Why Choose Tanishq Life Care for Pharmaceutical Export?
When it comes to reliable pharmaceutical export from India, Tanishq Life Care stands as a trusted name in the industry.
Key Strengths of Tanishq Life Care:
- High-quality pharmaceutical and nutraceutical products
- Compliance with international standards
- Expertise in third-party manufacturing and exports
- Strong quality control systems
- Competitive pricing for global markets
- Timely delivery and logistics support
Tanishq Life Care is committed to helping businesses expand globally by providing reliable and efficient export solutions.
Benefits of Pharmaceutical Export Business
Entering the pharmaceutical export business offers multiple advantages:
- High profit margins
- Global market reach
- Growing demand
- Long-term business sustainability
With the right strategy and partner, pharmaceutical export from India can become a highly profitable venture.
Future of Pharmaceutical Export from India
The future looks promising for Indian pharmaceutical exports.
Key Trends:
- Increased global demand for generics
- Growth in nutraceutical exports
- Expansion into regulated markets
- Digital transformation in supply chains
India is expected to remain a dominant player in the global pharmaceutical industry for years to come.
Conclusion
Pharmaceutical export from India offers immense opportunities for businesses looking to expand globally. With strong manufacturing capabilities, cost advantages, and global recognition, India continues to lead the pharmaceutical export market.
However, success in this industry requires a clear understanding of the export process, regulatory compliance, and market dynamics. Partnering with a reliable company like Tanishq Life Care can help you navigate these complexities and achieve sustainable growth.
If you are ready to enter the global pharmaceutical market, now is the perfect time to leverage India’s strengths and build a successful export business.
FAQ Section
What is the pharmaceutical export from India?
It refers to exporting medicines and pharmaceutical products manufactured in India to international markets.
Is pharmaceutical export from India profitable?
Yes, due to high global demand and cost advantages, it is a highly profitable business.
What licenses are required for pharma export?
You need an Import Export Code (IEC), drug license, and relevant certifications.
Which countries import pharmaceuticals from India?
USA, Europe, Africa, Southeast Asia, and the Middle East are major importers.
Can small businesses start pharmaceutical export?
Yes, with third-party manufacturing and proper licensing, even small businesses can enter the export market.